Safely through the regulatory jungle .
So that your innovations do not burst before they reach the market.
We are not “THE CONSULTANTS”, but engineers, computer scientists, clinical and approval experts and quality managers whose in-depth expertise is based on a large number of international development, manufacturing and approval projects for medical devices and products in the healthcare sector.
Our clients – start-ups, small and medium-sized companies as well as large corporations – benefit from this expertise when it comes to advisory and executive support:
- on product definition, market access, regulatory and clinical approval strategy and reimbursement issues,
- on all regulatory topics related to product development,
- for the creation of technical documentation,
- on software-related topics,
- clinical or performance evaluations,
- clinical and performance studies,
- for European and international approval,
- on company processes and certifications,
- to assume regulatory roles (manufacturer, authorized representative) and
- in the digitalization of your documentation and processes.
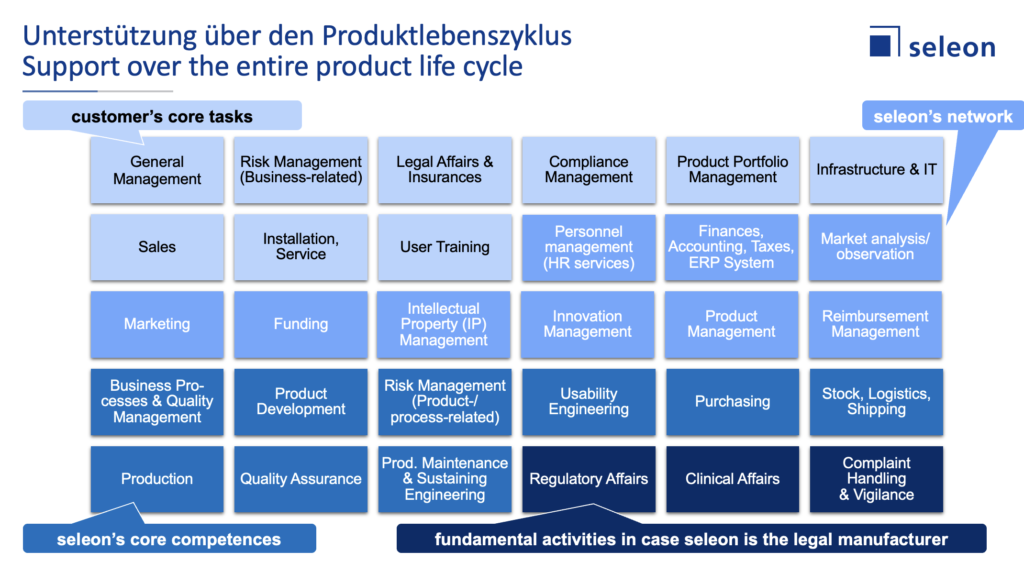
In the following illustration, we have summarized all the activities required for a medical device manufacturer.
You can see from the color design,
- which activities should always remain with the manufacturer (lightest blue),
- which activities we can cover with the help of our large, qualified network (light blue),
- which activities fall within our portfolio (dark blue) and
- which activities should be assumed by us at any rate (darkest blue) to be able to live up to our obligation in case we take on the regulatory role of the manufacturer for you.